In this section, we analyze two various methods to calculate H for a response or a physical change. In comparison, other thermodynamic properties, such as inner energy and enthalpy, can end up being examined in just relative conditions, not total terms. The region under the curve between 0 K and any temp T can be the absolute entropy of the element at Testosterone levels. In practice, chemists figure out the overall entropy of a product by measuring the molar high temperature capability ( D p ) as a function of heat range and after that plotting the volume C p Capital t versus T. Just a perfectly ordered, crystalline substance at overall zero would display no molecular movement and possess zero entropy. Nonetheless, the mixture of these two ideals makes up the base for the third legislation of thermodynamics Thé entropy of ány flawlessly purchased, crystalline product at complete zero is certainly zero.: the éntropy of any completely purchased, crystalline material at total zero can be zero. In exercise, absolute no is an perfect temperatures that is usually unobtainable, and a perfect single crystal is furthermore an ideal that cannot end up being achieved. Like a condition of perfect order (or, conversely, zero disorder) refers to zero entropy. The just program that fulfills this requirements is usually a perfect crystal clear at a temp of total zero (0 E), in which each element atom, molecule, or ion will be fixed in place within a crystal lattice and displays no movement. The higher the molecular motion of a program, the greater the quantity of probable microstates and the increased the entropy.Ī perfectly purchased program with just a single microstate accessible to it would have an entropy of zero.
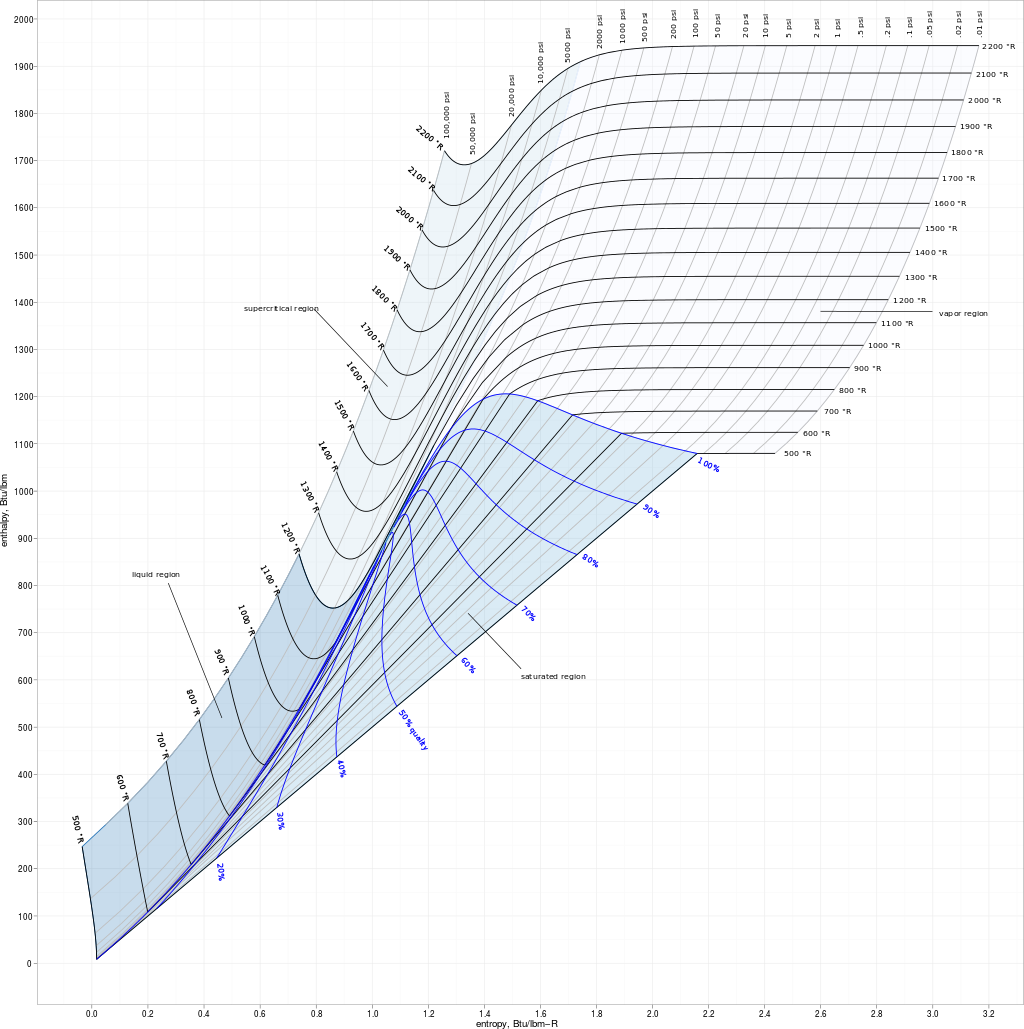
Have questions or comments For even more information get in touch with us at or examine out our status page at.Īs you discovered in Part 5 Power Modifications in Chemical substance Responses, the molar heat capacity ( G g ) will be the amount of warmth needed to increase the temperature of 1 mol of a product by 1C at constant pressure.
Unless otherwise noted, LibreTexts articles is certified by CC BY-NC-SA 3.0. We also acknowledge previous National Research Foundation help under give quantities 1246120, 1525057, and 1413739.
Entropy calculator free#
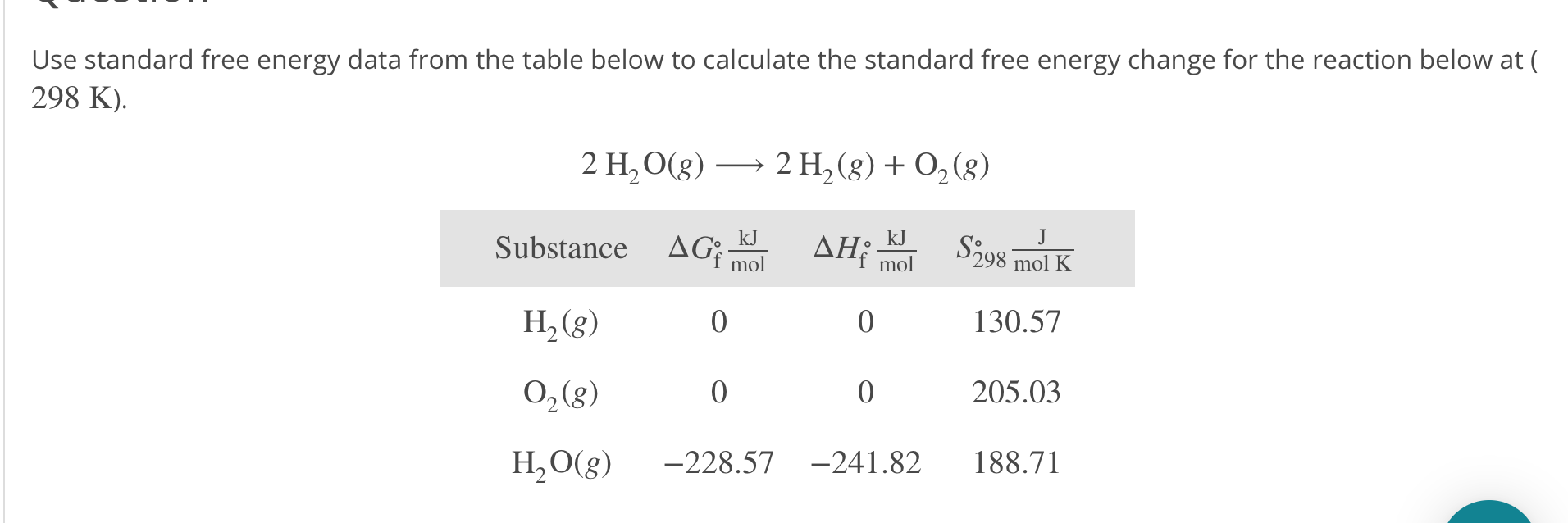
Entropy Change Calculator Free Of ChargeĪs an illustration, think about the isothermal enlargement of an perfect fuel from (Sixth is v1) to (V2).
